It is a rare but delightful opportunity to learn about something from an acknowledged world expert. Such was the case last month when I heard Mary-Claire King give the Stanley J. Korsmeyer Memorial lecture, hands-down one of the best talks I’ve ever heard. She was a wonderful public speaker: funny, charming, and straight-shooting.
Her topic, of course, was inherited breast and ovarian cancer. If you don’t know the story already, Dr. King wrote a wonderful perspective in Science about her role in the discovery of the BRCA1 gene and the race to clone it in the early 1990’s. Fascinatingly, she walked us through some of the pedigrees from early-onset breast cancer families described in the 1990 linkage study by her group.
The women in those families got breast cancer very young (20s or 3os) and usually died from it. Male obligate carriers were generally unaffected. Even for a highly penetrant mutation like BRCA1, there were exceptions, like the carrier who lived to 81 without ever getting cancer.
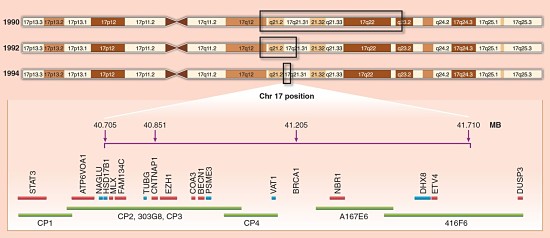
Of the seven early-onset breast cancer families, six harbored mutations in BRCA1 and one had a mutation in BRCA2. That paper was the culmination of 17 years of work and mapped the BRCA1 locus to chromosome 17.
The existence of a gene for predisposition to breast cancer triggered enormous interest in big labs in government, universities, and the private sector. It was the birth of cancer genetics.
BRCA1, DNA Repair, and Chemotherapy
At the time of its discovery, we knew nothing about the function of the BRCA1 gene. Subsequent genetics studies would reveal that it worked as a tumor-suppressor in a two-hit model of inherited cancer: the disease develops only after carriers of one loss-of-function mutation (generally a nonsense change or frameshift indel) lost the other copy to somatic mutation in a vulnerable cell type.
Normally, BRCA1 forms a heterodimer with BARD1, which stabilizes the BRCA1/BARD1/Fanconi complex. That complex repairs double-stranded DNA breaks via the homologous repair pathway. Mutations in several DNA repair genes — TP53, PALB2, CHEK2, BARD1, BRIP1, ATM, RAD51C, and RAD51D — are also known to predispose to breast and ovarian cancer.
Although BRCA1/2 carriers suffer a significantly higher risk of breast and ovarian cancer, they also tend to respond better to chemotherapy. This is not terribly surprising, because the loss of homologous DNA repair capability diminishes the ability of cancer cells to recover from DNA damage. Yet there’s also a different mechanism for DNA repair, non-homologous end joining (NHEJ), that does not involve BRCA1/2.
The bad news is that this may enable tumor cells to resist chemotherapy. The good news is that we have a class of drugs, PARP inhibitors, that block the NHEJ pathway. The first clinical trial of PARP inhibitors in BRCA1/2 null cancer patients “crashed,” according to Dr. King, because the compound being used didn’t actually inhibit PARP. New clinical trials are under way. Hopefully, they’ll demonstrate that PARP inhibitors make BRCA1/2 null patients more responsive to chemotherapy, which will make genetic testing even more critical.
Genetics and Epidemiology of Familial Breast Cancer
The epidemiology of breast cancer is fairly well known. By rough approximation, 1 in 8 women will get breast cancer at some point in her lifetime, and 10-20% of patients will turn out to carry an inherited mutation in a known predisposition gene. Like many cancers, risk of breast/ovarian cancer is highly age-dependent. BRCA1/2 carriers not only have a higher lifetime risk of disease, but also have a considerably higher age-dependent risk; some might even be diagnosed with disease in their 20’s or 30’s.
There is also a widely accepted trend related to breast cancer incidence that’s been apparent for decades: more women are getting it, and seemingly at younger ages. Indeed, Dr. King showed some results from two large epidemiological studies of breast cancer showing that the incidence curves (incidence by age, classified by carrier/non-carrier status) are quite striking if you segregate the women into two groups: those born before 1940, and those born after 1940.
There are lots of theories for why this might be, including some I might call conspiracy theories (e.g. radiation exposure, or hormones in milk). Yet Dr. King offered an explanation that I find both simple and convincing. We know that certain factors increase a woman’s risk of breast cancer. For example, the age of first menstruation (earlier = higher risk) and when she has her first child (later = higher risk).
In 1950, a woman typically began menstruating at 15 and bore her first child at 21. Today, menstruation often begins sooner (say age 11, due to some complicated factors like better nutrition) and the first child often comes later (age 30, because women often pursue higher education and/or careers).
Nutrition and education/independence, of course, are good things. However, the side effect is that the window of time between menstruation and first child went from ~6 years in 1950 to ~19 years today. And during that window, a woman’s breast tissues are bathed in estrogen. It makes for some super-healthy cells that don’t die easily, even if they suffer mutations. That longer window simply increases the odds that a second “hit” will occur in the gene for which a woman already carries a loss-of-function mutation.
In support of this idea, if researchers adjust for the length of that time window, the year-of-birth effect totally goes away. I think that’s some fascinating stuff.
Genetic Structure of BRCA1/2
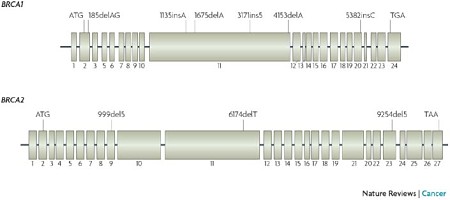
Interestingly, although the two most famous breast cancer susceptibility genes (BRCA1 and BRCA2) share no sequence similarity, they have a similar (and distinctive) genomic structure: many small exons and a large central exon. The central exon encodes a big portion of the protein and is surprisingly robust to amino acid substitutions, which is why most missense mutations in BRCA1 and BRCA2 are non-pathogenic.
Yet because these genes are so large, mutation databases have catalogued thousands of individual rare mutations that look deleterious. This is why a genotyping-based genetic test, like the one that was a cash cow for Myriad Genetics until recently, was never going to work in the long term. Now, with targeted sequencing, we have the capability to detect all types of mutation (substitutions, indels, even large SVs) affecting BRCA1/2 and other susceptibility genes.
From Gene Discovery to Population Screening
As the cost of sequencing-based genetic testing continues to drop, we’re in the position to screen the entire female population for cancer susceptibility genes.The World Health Organization offered guidelines for when genetic testing should be performed. In essence, four criteria must be met.
- The disease must be an important health problem
- Risk of disease for patients testing “positive” should be high.
- The mutations responsible for conferring risk must be identifiable
- Effective interventions must exist
Dr. King makes a pretty compelling argument that familial breast/ovarian cancer meets these requirements. #1 and #2 are well-established. #3 is true if you know your stuff: for a while, companies like Myriad leaned heavily on the “Variant of Unknown Significance” classification when they encountered a new variant, to the point that 88% of results were reported as such. Yet an expert team, like the one at UW, can classify all but <2% of variants as either pathogenic or non-pathogenic. The PARP inhibitor clinical trials should give us the answer for #4.
There are, of course, other considerations, like the cost of testing, the burden of genetic counseling, the age at which testing should be performed (Dr. King suggests 30), etc. Yet these are hurdles that can be overcome. Hurdles that must be overcome, if we’re to use our growing knowledge of disease genetics to improve the state of human health.
References
Hall JM, Lee MK, Newman B, Morrow JE, Anderson LA, Huey B, & King MC (1990). Linkage of early-onset familial breast cancer to chromosome 17q21. Science (New York, N.Y.), 250 (4988), 1684-9 PMID: 2270482
King MC (2014). “The race” to clone BRCA1. Science (New York, N.Y.), 343 (6178), 1462-5 PMID: 24675952